High School Science - HS-PS1-5 - How Temperature, Concentration, and Catalyst Affect Chemical Reaction Rates
6 Questions
NGSS.HS-PS1-5
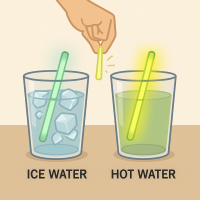
1
A student snaps two glow sticks of the same brand in two different cups of water.
2
Students test how a catalyst affects the rate of hydrogen peroxide decomposition (3% H₂O₂ at 25 °C).
3
Engineers explain that catalytic converters work better once the car engine warms up. Complete the sentences by using the drop-down menus.
4
A component of urban smog is the brownish gas nitrogen dioxide, which exists in equilibrium with its colorless dimer, dinitrogen tetroxide.
5
Part A: A student finds a brown plastic bottle of drugstore hydrogen peroxide (3%) in their bathroom cabinet.
6
Part B: Using your answers from Part A, explain why the hydrogen peroxide solution in the student’s bathroom cabinet became less effective over time.